Pancreatic Enzyme Replacement Therapy (PERT) – product brands: Creon®, Nutrizym® and Pancrex®.
Many of you are aware that there has been some variability and inconsistency in the delivery and availability, at local level, of PERT. This has led to some people either running out of or having difficulties/delays in getting their supply – particularly Creon® 25,000iu capsules.
Pancreatic enzymes are still being delivered regularly into the United Kingdom, but there have been temporary shortages and distribution issues with certain products and doses.
The shortages are a UK-wide problem, and we know other countries in Europe are also affected. Some areas may be more affected at different times – so you may or may not have any problems.
The company who produce Creon®, has assured us that although there has been a temporary shortage of Creon 25,000, their production plant is working to full capacity, with more than 150,000 packs of product being delivered to the UK every month.
The companies that make Nutrizym® and Pancrex® have also experienced shortages as people have switched to these brands to maintain their pancreatic enzyme replacement therapy, and they are working to keep up with the resulting increased demand.
We are collaborating with charity colleagues to identify where issues may be occurring.
This will help lead towards a solution and has included meetings with the suppliers, the NHS, Department of Health and Westminster – and reaching out to Medicine Teams across the devolved nations.
We welcome and fully support the Position Statement: Pancreatic enzyme replacement therapy (PERT) shortage – published 24th March 2024 – which includes advice for the management of adults with pancreatic exocrine insufficiency.
(Full document is available here).
This document is also endorsed by the British Society of Gastroenterology (Pancreas section); Pancreatic Society of Great Britain and Ireland, Pancreatic Cancer UK, GUTS UK, Cystic Fibrosis Trust, CF Medical Association and the British Dietetic Association.
The position paper is designed to provide advice to minimise the impact on your symptoms and quality of life should you be unable to access your normal supply of PERT.
We would like to thank authors Mary Phillips, Laura McGeeney, Jacqueline Lowdon & Kerry-Lee Watson for their expertise in providing this timely guidance.
- If you have cystic fibrosis, please contact the Cystic Fibrosis Trust, as some of the information will be different for you.
- If you have pancreatitis, please contact Guts UK.
- If you take PERT for other reasons, please contact your healthcare professional for advice.
We will update this information regularly as the situation changes.
We know this has been a worrying time – if you need help or information about how to deal with problems getting your PERT – we would suggest contacting your dietitian if you have one, GP or specialist nurse for support with digestion and PERT. If you are having any difficulty in getting hold of your specialist healthcare professionals, please do contact our Helpline or use our “Contact Us” form HERE and we will try to help.
Neuroendocrine Cancer UK Helpline (Tuesdays-Thursdays, 10am – 4pm): 0800 434 6476
The following advice, based on guidance from the Position Statement, is for people with neuroendocrine cancer that take PERT.
–

–
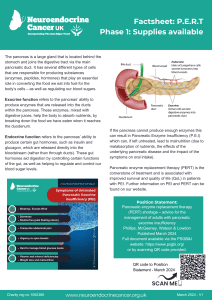
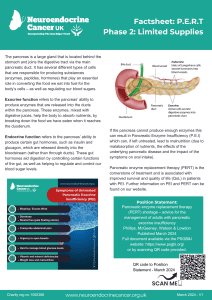
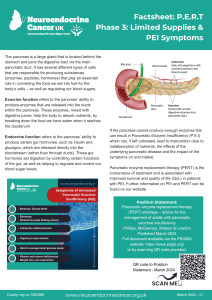
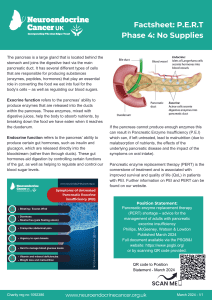
Pancreatic enzyme replacement therapy is prescribed to support adequate digestion in people
with primary or secondary pancreatic exocrine insufficiency (PEI), most commonly due to either pancreatic neuroendocrine cancer or those with neuroendocrine cancer that take somatostatin analogues (e.g. Octreotide or Lanreotide). Other neuroendocrine cancer treatments, including pancreatic surgery, PRRT and/or chemotherapy may also affect pancreatic function – either temporarily or more long-term.
There are many other clinical situations where people may have primary or secondary PEI, such as pancreatic adenocarcinoma (PDAC), pancreatitis, cystic fibrosis, type 3c diabetes or following gastrectomy or gastric bypass surgery.
Regardless of the cause of the PEI, the types of symptoms may be similar, but their severity can vary from person to person.
These symptoms are usually treated by taking PERT and may recur/increase if you are unable to take enough. Brands of PERT in the UK are Creon®, Nutrizym® and Pancrex®.
If you have enough supply and your symptoms are well treated and weight stable – we recommend that you take your PERT as advised by your dietitian, specialist nurse and/or medical team.
Phillips et al (2021) Consensus for the management of pancreatic exocrine insufficiency: UK practical guidelines. BMJ Open Gastroenterology
Patient information video on PERT available HERE
Please note the following advice is designed for adults with PEI, specialist advice should be sought for children with PEI.
The Position Statement divides advice for patients into 4 phases, depending on the supply available:
 .
.  .
. 
- Phase 1 – Supplies available
- Phase 2 – Limited supplies
- Phase 3 – Insufficient supplies and you have abdominal symptoms
- Phase 4 – No supplies available
Position Statement: Pancreatic enzyme replacement therapy (PERT) shortage – published 24th March 2024 – which includes advice for the management of adults with pancreatic exocrine insufficiency.
